Axis 1: Impact of metabolic state on multisensory signaling.
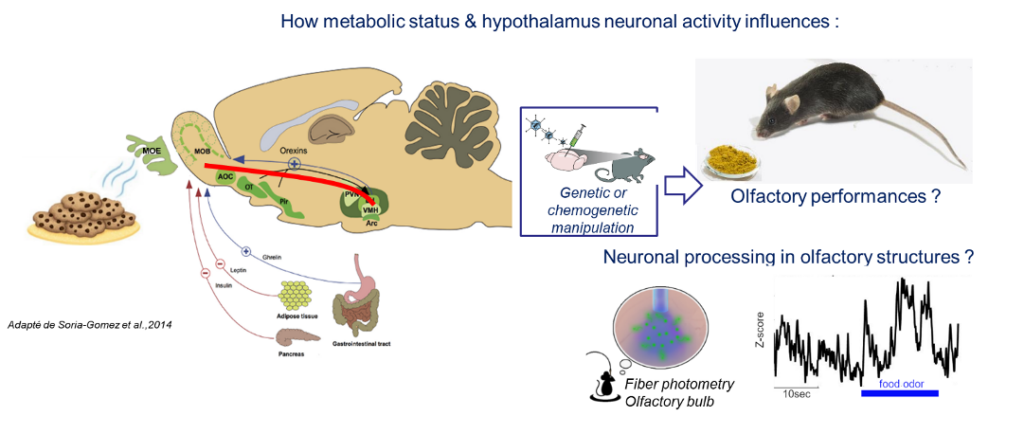
Food is a common multisensory experience, where flavor is a combination of smell, taste, and touch. Smell is one of the most relevant sensory cues predicting food and is likely to play a key role in food choice and consumption. In addition, most odorants also activate the trigeminal nerve, which is part of the touch pathway. It is currently unknown whether functional changes in the trigeminal activation could be involved in the alteration of olfaction in obese individuals. Evidence shows that the olfactory system is at the crossroads between sensory processing and metabolic sensing (Faour et al., 2022, Neuropharmacology 206, 108923). The olfactory bulb detects and is modulated by the main hormones and nutrients involved in the regulation of food intake. However, we still ignore the impact of the brain region that has a major control on food intake, the hypothalamus, on olfactory activity and olfactory driven feeding behavior. Our hypothesis is that the hypothalamo-olfactory network modulates the quality and/or the reward value of an odorant according to the internal state.
We combine behavioral and genetic approaches (transgenic mice, Designer Receptors Exclusively Activated By Designer Drugs) and imaging of fluorescent biosensors (GCaMP-based fiber photometry) in behaving mice to assess the impact of AgRP-neurons manipulation or the impact of trigeminal nerve inhibition on olfactory bulb neuronal activity and olfactory behavior.
Fundings: ANR Idex Emergence, Cooperation CNRS-UChicago (80Prime PhD fellowship), ANR HypoCamK, METABRAIN
Human resources: Claire Martin (CM), Maya Four (MF), Julien Castel (JC), Serge Luquet (SL), Giuseppe Gangarossa (GG)
Axis 2: Astrocytic control of metabolism and reactivity in response to obesity
Nutrient overload and obesity promote maladaptive changes in astrocytes physiology, thereby leading to impaired central control of food intake, energy expenditure and glucose metabolism (Garcia-Caceres et al., 2019, Nature neuroscience 22, 7-14.). We have characterized alterations of the structural and functional properties of astrocytes and astrocyte-neuron communication and discovered that hypothalamic astrocytes in the paraventricular nucleus exert a direct control on body weight regulation and glucose metabolism (Herrera Moro Chao et al., 2022 Cell Metab 34, 1532-1547 e1536)).
In the striatum, we showed that activation of astrocytes of the dorsal striatum restored obesity-induced cognitive defect and led to a reinstatement of striatal neuronal synchronicity. On the other hand, activation of astrocytes in the Nucleus Accumbens altered whole-body substrate utilization and energy expenditure (Montalban et al, BioRxiv).
We want to go further in the examination of the astrocyte cellular and molecular profile adaptive response to DIO. We study the impact of astrocyte-astrocyte and astrocyte-neurons communication in vivo using pharmacological and genetic tools including DREADD/chemogenetic manipulation of Gq/Gi signaling. These tools are coupled with in vivo recording of metabolic efficiency and astrocytic and neuronal activity (GCaMP based fiber photometry).
Fundings : ANR AstrObesity.
Human ressources: Serge Luquet (SL), Claire Martin (CM), Caroline Leger (CL), Julien Castel (JC), Anthony Ansoult (AA), Giuseppe Gangarossa (GG) + partners
Axis 3: Molecular determinants of brain susceptibility/resilience to feeding & metabolic diseases.
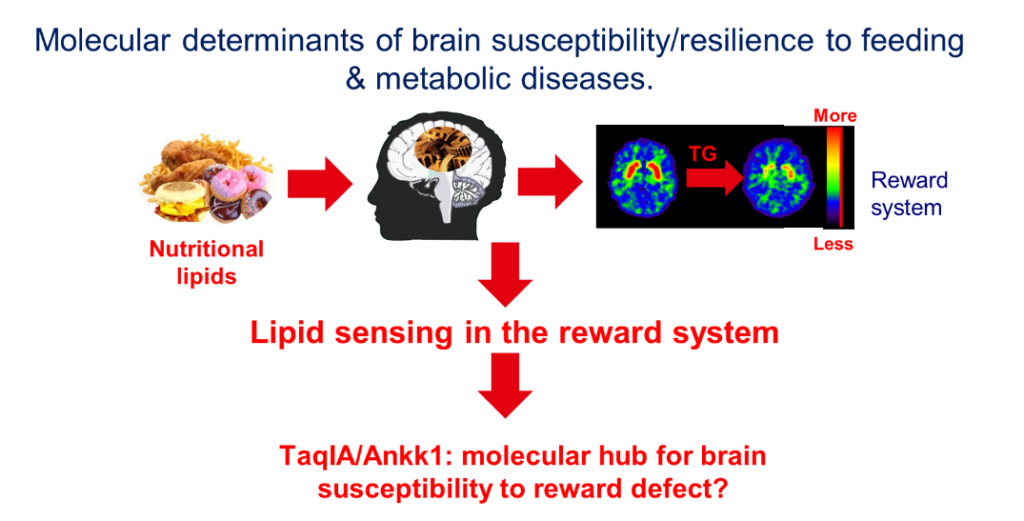
Introduction. We have published seminal studies showing that circulating triglycerides (TG) can be metabolized within the dopamine (DA) reward system and modulate reinforced behaviors by gating the activity of DA subtype 2 receptor (D2R)-expressing neurons (Berland et al., 2020, Cell Metab 31, 773-790 e711.; Berland et al., 2021, Trends Endocrinol Metab 32, 693-705.; Cansell et al., 2014, Molecular psychiatry 19, 1095-1105.). Moreover, in humans, we observed that post-prandial TG excursions correlated with food-cues-elicited brain activities in individuals carrying the genetic risk factor TaqIA, the latter corresponding to altered D2R signaling. Collectively, these findings unveil a novel mechanism by which dietary TG directly alter signaling events, thereby providing a new mechanistic basis by which energy-rich diets lead to (mal)adaptations that may underlie reward deficits and compulsive behaviors (Berland et al., 2020, Cell Metab 31, 773-790 e711.; Sun et al., 2017, Trends Cogn Sci 21, 372-384.). Our goal is now to (1) explore the cellular and molecular mechanisms by which circulating lipids alter DA transmission and (2) provide a molecular mechanism by which TaqIA/Ankk1 polymorphism drives susceptibility to metabolic defects.
Methodology. Brain lipid sensing and reward. We will use both brain-specific TG delivery (BTGD) in lean and obese mice to explore how the obesogenic environment affects the function and excitability of DA circuits in response to food-cues combined with in vivo recordings (fiber photometry) of DAergic and D2R-expressing neurons using DA and Ca2+ biosensors. We are assessing how brain lipid sensing affects food choice, food reinforcement and metabolic efficiency (Berland et al., 2020, Cell Metab 31, 773-790 e711.; Berland et al., 2021, Trends Endocrinol Metab 32, 693-705) in lean and obese mice. At the molecular level, we will explore cell-type specific transcriptomic alterations following BTGD and/or diet-induced obesity (DIO) using TRAP methods to specifically capture actively translating mRNA in D2R-expressing neurons and assess how high-fat mediates transcriptional adaptations. TaqIA/Ankk1 polymorphism and reward dysfunction. The TaqIA A1 single nucleotide polymorphism (SNP, rs1800497) allele is located in the gene that codes for the Ankyrin repeat and kinase domain containing 1 kinase (ANKK1) near the Drd2 gene. The TaqI SNP is the most studied polymorphism in psychiatric diseases but aside from correlative studies in humans, there is no mechanism described as to the role of ANKK1 (review see (Sun et al., 2017, Trends Cogn Sci 21, 372-384)). We have developed an Ankk1fl/fl mouse line and studied the consequence of Ankk1 ablation in the reward circuits. We discovered that Ankk1 controls reward and metabolism through the regulation of D2R-neurons activity (Montalban. E et al. 2023. Bio Psy). In collaboration with BFA team 6 and HMGU (A. Serrano, HMGU, Munich) we have the tools to identify potential molecular mechanisms by which A1 and A2 variants of Ankk1 might affect neural activity.
Expected Results. 1-Characterizing and identifying the molecular adaptations triggered by chronic exposure to TG within the reward system. 2-Determining whether and how dietary TG can alter the function and excitability of mesolimbic neural circuits. 3-Determining the role of Ankk1 in Mesolimbic/Hypothalamic neural circuit adaptation. 4-Determining the molecular basis by which Ankk1 A1/A2 alters D2R/DA signaling.
Fundings: This project is primarily funded through the MDPRC grant, the FRM team 2020, FRC Luquet, Allen Foundation, Nutricia Research Foundation, and partly through the ANR Rewan and Bariatek funding. Institutional support to set-up US-France collaborative network is funded through the CNRS International Research Project (IRP) BRAINHEALTH.
Human resources: Serge Luquet (SL), Giuseppe Gangarossa (GG), Claire Martin (CM), Julien Castel (JC), Guangping Li (GL), Anthony Ansoult (AA), Fanny Bain (FB), Caroline Leger (CL), Nour Mesto (NMe), Claudia Grajeda (CG) + partners.
Axis 4: Interoceptive dynamics controlling homeostatic and reward processes
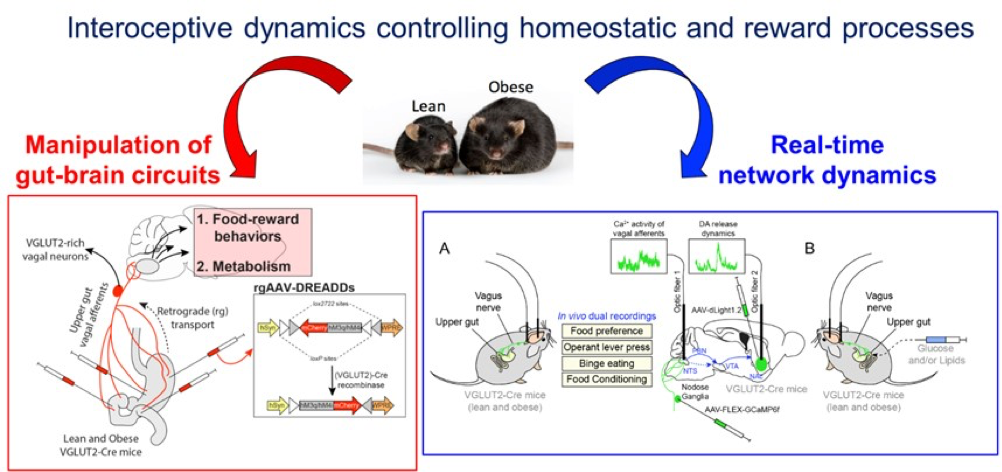
Introduction. Interoceptive signals arising from the periphery, through the vagus nerve, constantly inform the brain regarding the physiological states of peripheral organs, therefore scaling the activity of brain circuits and influencing the elaboration of brain-orchestrated adaptive strategies. We have recently found that ambient peripheral endocannabinoids (eCBs) bidirectionally gate the activity of the gut-brain vagal axis (Berland et al., 2022, Mol Psychiatry 27, 2340-2354.) and contribute to the establishment of reward-based eating disorders. Moreover, we have also reported that interoceptive and homeostatic information can be relayed from downstream (hindbrain/brainstem) to upstream brain regions by mobilizing the dense hub of the paraventricular thalamus (PVT) (Dumont et al., 2022 JPhysiol). Our current hypothesis is that the maladaptive plasticity of the vagus nerve and the distortion of the interoceptive trajectories can lead to the dysregulation of the neural centers of the “extended” gut-brain axis and promote eating disorders (i.e., obesity).
Methodology. First, we will assess whether and how obesity functionally disrupts the interconnected neuronal networks and dynamics allowing the integration of interoceptive information and the elaboration of (mal)adapted responses. We will mainly study novel interoceptive gut-brain trajectories which ultimate in the regulation of food valence (i.e. reinforcement, reward), notably the vagus nervebrainstem/hindbrainNAc pathway and its intermediate PVT-dependent hub. By mobilizing projections- and cell type-specific approaches, we will use in vivo bulk fiber photometry to simultaneously record the Ca2+ activity and the releasing dynamics of mediators (dopamine, acetylcholine, GLP-1, glutamate) at the level of multiple segments of the gut-brain axis. The interoceptive trajectories will be depicted during spontaneous/evoked feeding, food preference, food-motivated behaviors and intragastric delivery of nutrients in lean and obese mice. Next, we will use networks-restricted microinjections of Cre/Flp-dependent viruses expressing engineered receptors (Gq, Gi, NaChBac, Kir2.1) to manipulate the neuronal components of the gut-brain axis in reward feeding processes, metabolism and body weight gain. Again, we will focus our functional analysis at the network level: vagus nervebrainstem/hindbrainNAc pathway and its intermediate PVT-dependent hub. Finally, at the molecular level we will explore how the obesogenic environment leads to transcriptomic (mal)adaptations of interconnected gut-to-brain structures using projections and/or cell type-specific methods.
Expected Results. This work will unravel the circuits as well as the cellular and molecular mechanisms by which gut-generated and vagus-mediated distorted interoceptive inputs contribute to the (dys)regulation of food-reward eating disorders, body weight maintenance and energy metabolism.
Fundings: This project is primarily funded through the ANR JCJC HERO, ANR ParObesity, FRC-Gangarossa, Nutricia Research Foundation, IReSP, Idex Emergence ThalHOME, IUF, FRM and partly through the Bariatek funding.
Human resources: Giuseppe Gangarossa (GG), Oriane Onimus (OO), Benoit Bertrand (BB), Nejmeh Mashhour (NMa), Camille De Almeida (CdA), Julien Castel (JC), Serge Luquet (SL), Claire Martin (CM) + partners